 If lone pairs are absent in the compounds then the molecular geometry matches with the geometry of electronics and is octahedral.For example, sulphur hexafluoride (SF6) where S (sulphur) is the central atom and F (fluorine)is the atom or ligand attached to the central atom. Where X is a central metal and F is the ligands attached to the central atom to form the octahedral shape. The octahedral shape of molecules has a generic formula which is XF6.When more than one ligand or we can say atoms are attached to the centre of an octahedral atom, then the complex which is formed by the attachment of these atoms can also exist in isomers.Octahedral shape of molecules has a steric number which is six. As we all know that a perfect octahedron belongs to the point ‘Oh’. Molecules that have octahedral structures have square planar geometry. All the bonds in which an octahedral shape of molecules have made a 90-degree angle with each other or in other words it have a bond angle of 90 degrees. The arrangement of the surrounding atom has given octahedral molecules their overall shape of eight connected triangles. An example of octahedral compounds is molybdenum hexacarbonyl (Mo(CO)6). It has the prefix octa which means eight. It has two square pyramids back to back, each square pyramid with four faces. The Octahedral Shape of Molecules contains eight faces. doi: 10.1016/S0003-2670(96)00563-6.The octahedral shape of molecules is the shape of molecules where six atoms or ligands or groups of atoms are arranged in a systematic way around a central dogma or atom. "Xenon tetrafluoride as a decomposition agent for silicone rubber for isolation and atomic emission spectrometric determination of trace metals". Journal of the Chemical Society D: Chemical Communications (23): 1543–1544. "Trifluoroxenon(IV) µ-fluoro-bispentafluoroantimonate(V): the XeF +ģ cation". ^ Suzuki, Hitomi Matano, Yoshihiro (2001).^ Harding, Charlie Johnson, David Arthur Janes, Rob (2002)."Xenon Tetrafluoride: Reaction with Aqueous Solutions". Parsons, Roger Jordan, Joseph International Union of Pure and Applied Chemistry (1985). ^ a b c Haner, Jamie Schrobilgen, Gary J."Xenon Tetrafluoride Molecule and Its Thermal Motion: A Neutron Diffraction Study". "Xenon Tetrafluoride: Crystal Structure". "Xenon Tetrafluoride: Fluorine-19 High-Resolution Magnetic Resonance Spectrum". Translated by Eagleson, Mary Brewer, William. XeFĤ reacts with the silicone to form simple gaseous products, leaving a residue of metal impurities. It has been shown to degrade silicone rubber for analyzing trace metal impurities in the rubber. Xenon tetrafluoride has few applications. The reaction of xenon tetrafluoride with platinum yields platinum tetrafluoride and xenon: XeF 4 + Pt → PtF 4 + Xe Applications Reaction with bismuth pentafluoride ( BiFģ cation in the salt XeF 3Sb 2F 11 has been characterized by NMR spectroscopy. Reaction with tetramethylammonium fluoride gives tetramethylammonium pentafluoroxenate, which contains the pentagonal XeF −ĥ anion is also formed by reaction with caesium fluoride: CsF + XeF It is used as a precursor for synthesis of all tetravalent Xe compounds. Xenon tetrafluoride hydrolyzes at low temperatures to form elemental xenon, oxygen, hydrofluoric acid, and aqueous xenon trioxide. The low volatility of XeFĦ allows it to be purified by fractional sublimation. The nickel is not a catalyst for this reaction nickel containers are used because they react with fluorine to form a protective, non-peeling passivation layer of nickel(II) fluoride NiFĢ on their interior surfaces. Some xenon difluoride ( XeFĦ) is also produced, where increased temperature or decreased fluorine concentration in the input mixture favors XeFĢ production, and decreased temperature or increased fluorine concentration favors XeFĦ. Xenon tetrafluoride is produced by heating a mixture of xenon and fluorine in a 1:5 molar ratio in a nickel container to 400 ☌. According to VSEPR theory, in addition to four fluoride ligands, the xenon center has two lone pairs of electrons. 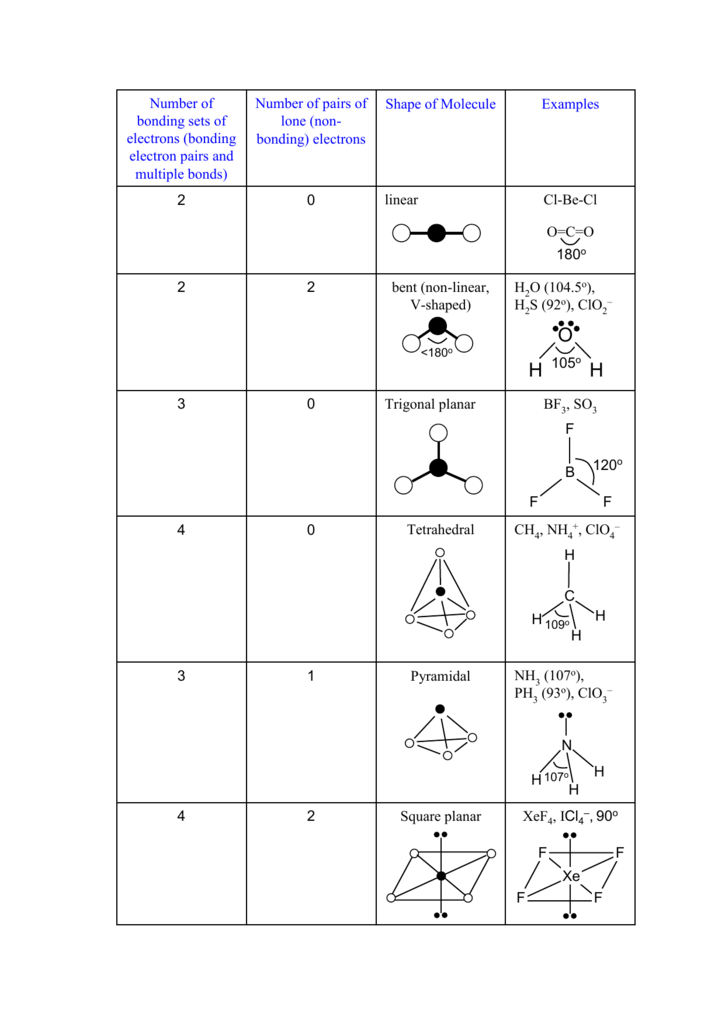
The structure is square planar, as has been confirmed by neutron diffraction studies. Its structure was determined by both NMR spectroscopy and X-ray crystallography in 1963. 
Xenon tetrafluoride is a colorless crystalline solid that sublimes at 117 ☌. This reaction is exothermic, releasing an energy of 251 kJ/mol. It is produced by the chemical reaction of xenon with fluorine: Xe + 2 F It was the first discovered binary compound of a noble gas. Xenon tetrafluoride is a chemical compound with chemical formula XeFĤ. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
